 After unbound antibodies are washed off, fluorescently labeled secondary antibodies, which bind to the primary antibodies, are added. Typically, primary antibodies, which recognize the proteins of interest in the cell, are first introduced. In immunofluorescence, the cells are first fixed to cross-link proteins in the cell and then permeabilized to allow antibodies access to the cellular milieu. The two major techniques for introducing fluorescent labels into cells are immunofluorescence, in which fluorescently labeled antibodies that bind to specific proteins in cells are introduced, and genetic introduction of a fluorescent protein. A fluorescence filter cube separates the light by wavelength so that the emitted light can be imaged without interference from the excitation light ( Murphy and Davidson, 2012). Fluorescence microscopy is typically done using epifluorescence, in which the fluorescence excitation light illuminates the sample through the same objective that is used to detect the emission from the sample. Multiple different fluorescent molecules can be distinguished simultaneously and can be detected at very low abundance (single molecules can be imaged), making this a very powerful technique. This allows the labels to be targeted to the molecule(s) of interest, either by genetically encoding a fluorescent protein or by binding a fluorescently labeled antibody. Most molecules in the cell are not very fluorescent, so fluorescent labels to be imaged are typically introduced by the experimenter. Brightfield without phase contrast or DIC is usually sufficient to see the general outlines of cells, but phase contrast or DIC is necessary to achieve detailed, high-contrast brightfield images.įluorescence microscopy uses fluorescent dyes (fluorophores), which are molecules that absorb one wavelength of light (the excitation wavelength) and emit a second, longer wavelength of light (the emission wavelength). The two most commonly used techniques to visualize this phase shift are phase contrast, which causes cells to appear dark on a light background, and differential interference contrast (DIC), which gives a pseudo–three-dimensional (3D) shaded appearance to cells ( Murphy and Davidson, 2012). 
Because most cells are thin and transparent, they do not absorb much light and so are difficult to see without adding optics that allows the phase shift of light induced by the cells to be seen. 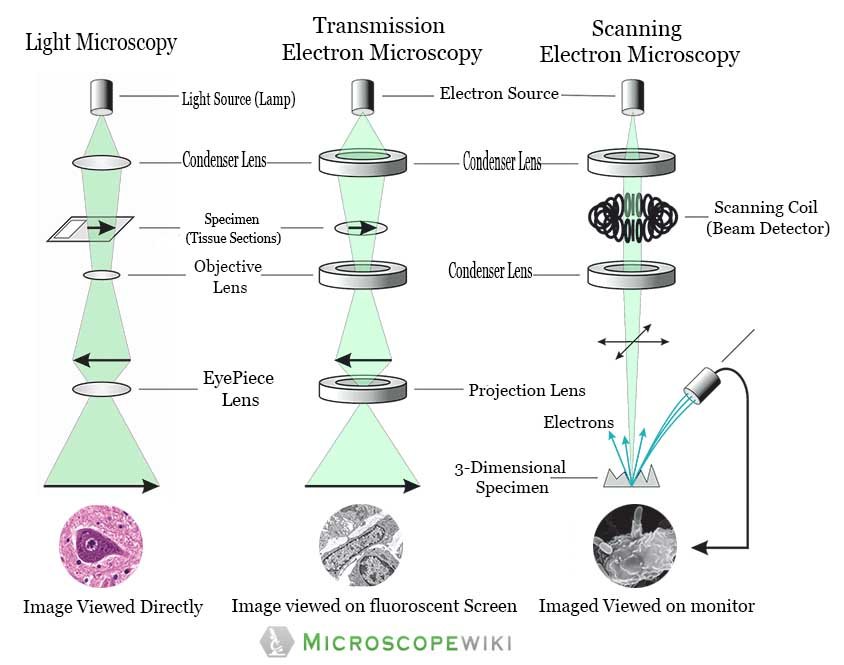
In brightfield microscopy, the light source and detection objective are placed on opposite sides of the sample, and the sample is imaged by its effect on the light passing through it as the sample absorbs, scatters, or deflects the light. In this protocol, we suggest a combination of CLEM and 3D electron microscopy to perform detailed structural studies of mitochondria and ER.Most broadly, light microscopy techniques can be divided into two categories: brightfield and fluorescence. An en bloc staining method, ultrathin serial sectioning (array tomography), and volume electron microscopy were applied to observe the 3D structure. Therefore, we observed the morphology of mitochondria and ER via correlative light-electron microscopy (CLEM) and volume electron microscopy techniques using enhanced ascorbate peroxidase 2 and horseradish peroxidase staining. Transmission electron microscopy provides good membrane contrast and nanometer-scale resolution for the observation of cellular organelles however, it is exceptionally time-consuming when assessing the three-dimensional (3D) structure of highly curved organelles. Visualization of this network between mitochondria and ER has been attempted using super-resolution fluorescence imaging and light microscopy however, the limited resolution is insufficient to observe the membranes between the mitochondria and ER in detail. These highly curved structures are folded into various shapes to form a dynamic network depending on the cellular conditions. 
Cellular organelles, such as mitochondria and endoplasmic reticulum (ER), create a network to perform a variety of functions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
